Page 141 - Chemistry ICSE Class X
P. 141
Mole Concept and Stoichiometry 127
12
Mass of a C atom
1 atomic mass unit = 6
12
12
Absolute mass of a C atom is 1.9924 × 10 –23 g. Therefore,
6
1.9924 × 10 –23 g
1 atomic mass unit = = 1.66 × 10 –24 g = 1.66 × 10 –27 kg
12
What is the atomic mass (A) of an element
The average mass of an atom of an element is known as its atomic mass.
Atomic mass of an element is designated as A. Since, atomic mass is actual dŚĞ ĂƚŽŵŝĐ ŵĂƐƐ ƵŶŝƚ ŝƐ ŶŽƚ Ă
mass, hence it has the unit of mass, viz., gram, kilogram, atomic mass unit, ƉƌĂĐƟĐĂů ƵŶŝƚ ďĞĐĂƵƐĞ ŝƚ ŝƐ ĞƋƵŝǀĂůĞŶƚ
ƚŽ Ă ǀĞƌLJ ƐŵĂůů ŵĂƐƐ͘ KŶĞ ĂƚŽŵŝĐ
etc. The internationally accepted unit for describing atomic mass is atomic ŵĂƐƐ ƵŶŝƚ ŝƐ ĞƋƵĂů ƚŽ ϭ͘ϲϲ п ϭϬ ʹϮϳ ŬŐ͕
mass unit. The average mass of an atom of an element in atomic mass unit Žƌ ϭ͘ϲϲ п ϭϬ ʹϮϰ Ő͘ ^ƵĐŚ ƐŵĂůů ŵĂƐƐĞƐ
is called its atomic mass. ĐĂŶŶŽƚ ďĞ ŵĞĂƐƵƌĞĚ ŽŶ ĂŶLJ ďĂůĂŶĐĞ͘
ͻ
What is relative atomic mass (A ) of an element WůĞĂƐĞ ŶŽƚĞ ƚŚĂƚ ƚŚĞ ŵĂŐŶŝƚƵĚĞ ŽĨ
r
The relative atomic mass (A QH CP GNGOGPV KU FGſPGF CU VJG CXGTCIG OCUU ƚŚĞ ƌĞůĂƟǀĞ ĂƚŽŵŝĐ ŵĂƐƐ ĂŶĚ ƚŚĞ
r
12
of an atom of the element compared with an atom of C taken as 12 u. ĂƚŽŵŝĐ ŵĂƐƐ ŽĨ ĂŶ ĞůĞŵĞŶƚ ĂƌĞ ƚŚĞ
6
ƐĂŵĞ͕ ďƵƚ ƚŚĞ ƚǁŽ ĚŝīĞƌ ŝŶ ƵŶŝƚƐ͘
Thus, ͻഩ dŚĞ ƌĞůĂƟǀĞ ĂƚŽŵŝĐ ŵĂƐƐ ŚĂƐ ŶŽ
Average mass of 1 atom of the element ƵŶŝƚ͘
Relative atomic mass of an element (A ) =
r
12
(Mass of one C atom)/12 ͻഩ dŚĞ ĂƚŽŵŝĐ ŵĂƐƐ ŝƐ ĞdžƉƌĞƐƐĞĚ ŝŶ
6
The relative atomic mass is denoted by A . The relative atomic mass is ƚŚĞ ĂƚŽŵŝĐ ŵĂƐƐ ƵŶŝƚƐ͘
r
a pure number, and hence it has no unit.
The relative atomic mass of an element indicates the number of times
12
one atom of that element is heavier than th of a C atom. For example,
1
6
12
the average relative mass of chlorine is 35.45. This means that an atom of
12
chlorine on average is 35.45/12 times heavier than one atom of C.
6
The names, symbols and relative atomic masses of some common
elements are presented in Table 5.1.
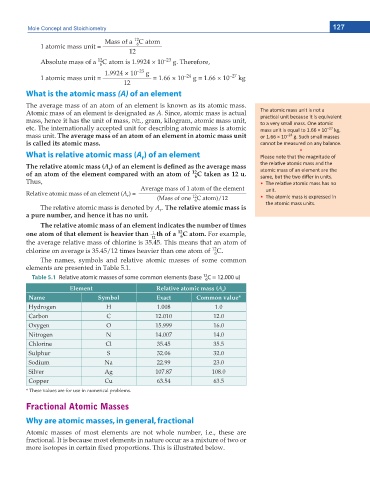
12
Table 5.1 Relative atomic masses of some common elements (base C = 12.000 u)
6
Element Relative atomic mass (A )
r
Name Symbol Exact Common value*
Hydrogen H 1.008 1.0
Carbon C 12.010 12.0
Oxygen O 15.999 16.0
Nitrogen N 14.007 14.0
Chlorine Cl 35.45 35.5
Sulphur S 32.06 32.0
Sodium Na 22.99 23.0
Silver Ag 107.87 108.0
Copper Cu 63.54 63.5
* These values are for use in numerical problems.
Fractional Atomic Masses
Why are atomic masses, in general, fractional
Atomic masses of most elements are not whole number, i.e., these are
fractional. It is because most elements in nature occur as a mixture of two or
OQTG KUQVQRGU KP EGTVCKP ſZGF RTQRQTVKQPU 6JKU KU KNNWUVTCVGF DGNQY